|
3/24/2023 0 Comments Carbon dating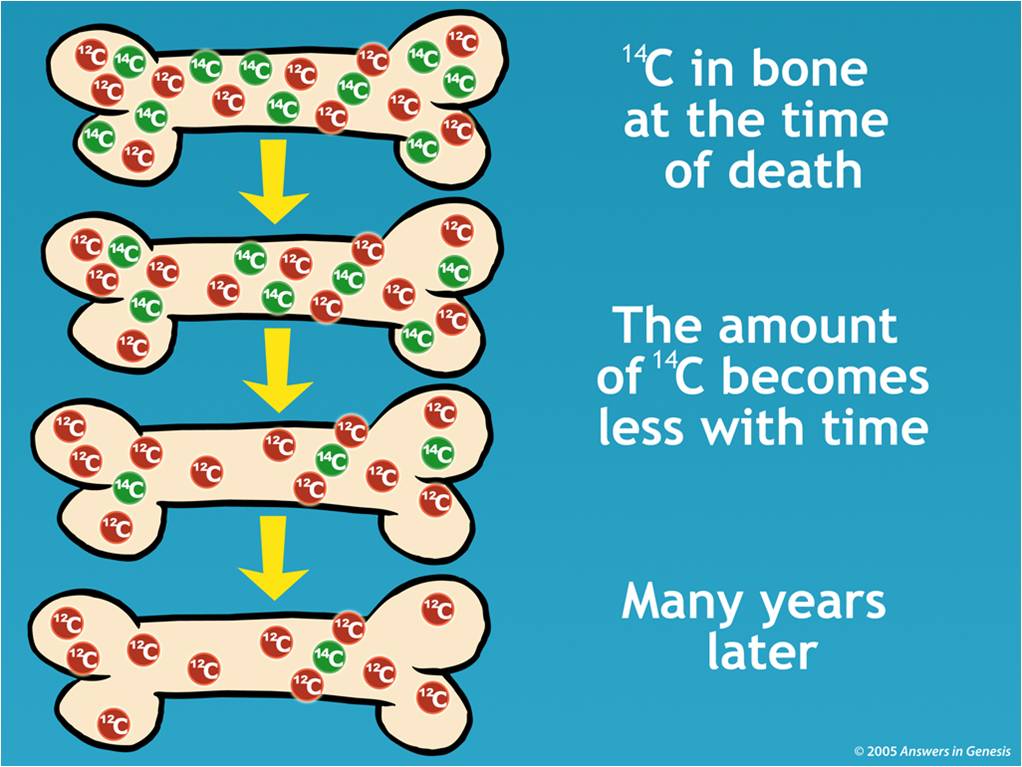
Together carbon –12 and carbon –13 make up 99% of all naturally occurring carbon. The most abundant isotope in nature is carbon –12, followed in abundance by carbon –13. Carbon has isotopes with atomic weights between 9 and 15. He became intrigued by carbon –14, a radioactive isotope of carbon. The first method for dating organic objects (such as the remains of plants and animals) was developed by another American chemist, Willard Libby (1908 –1980). While the uranium-lead dating method was limited (being only applicable to samples containing uranium), it was proved to scientists that radioactive dating was both possible and reliable. Boltwood used this method, called radioactive dating, to obtain a very accurate measurement of the age of Earth. Thus, the greater the amount of lead, the older the rock. This was because uranium, as it underwent radioactive decay, would transmute into lead over a long span of time. In 1907, the American chemist Bertram Boltwood (1870 –1927) proposed that rocks containing radioactive uranium could be dated by measuring the amount of lead in the sample. However, many objects were found in caves, frozen in ice, or in other areas whose ages were not known in these cases, it was clear that a method for dating the actual object was necessary. By comparing the placement of objects with the age of the rock and silt layers in which they were found, scientists could usually make a general estimate of their age. It is based on the decay rate of the radioactive carbon isotope 14C, a form of carbon taken in by all living organisms while they are alive.īefore the twentieth century, determining the age of ancient fossils or artifacts was considered the job of paleontologists or paleontologists, not nuclear physicists. This article appeared in NIJ Journal Issue 269, March 2012, as a sidebar to the article Applying Carbon-14 Dating to Recent Human Remains by Philip Bulman with Danielle McLeod-Henning.Carbon dating is a technique used to determine the approximate age of once-living materials. Using biological knowledge of the timing of a particular tissue's formation, one calculates the year of birth or death of the organism from which the tissue was derived. 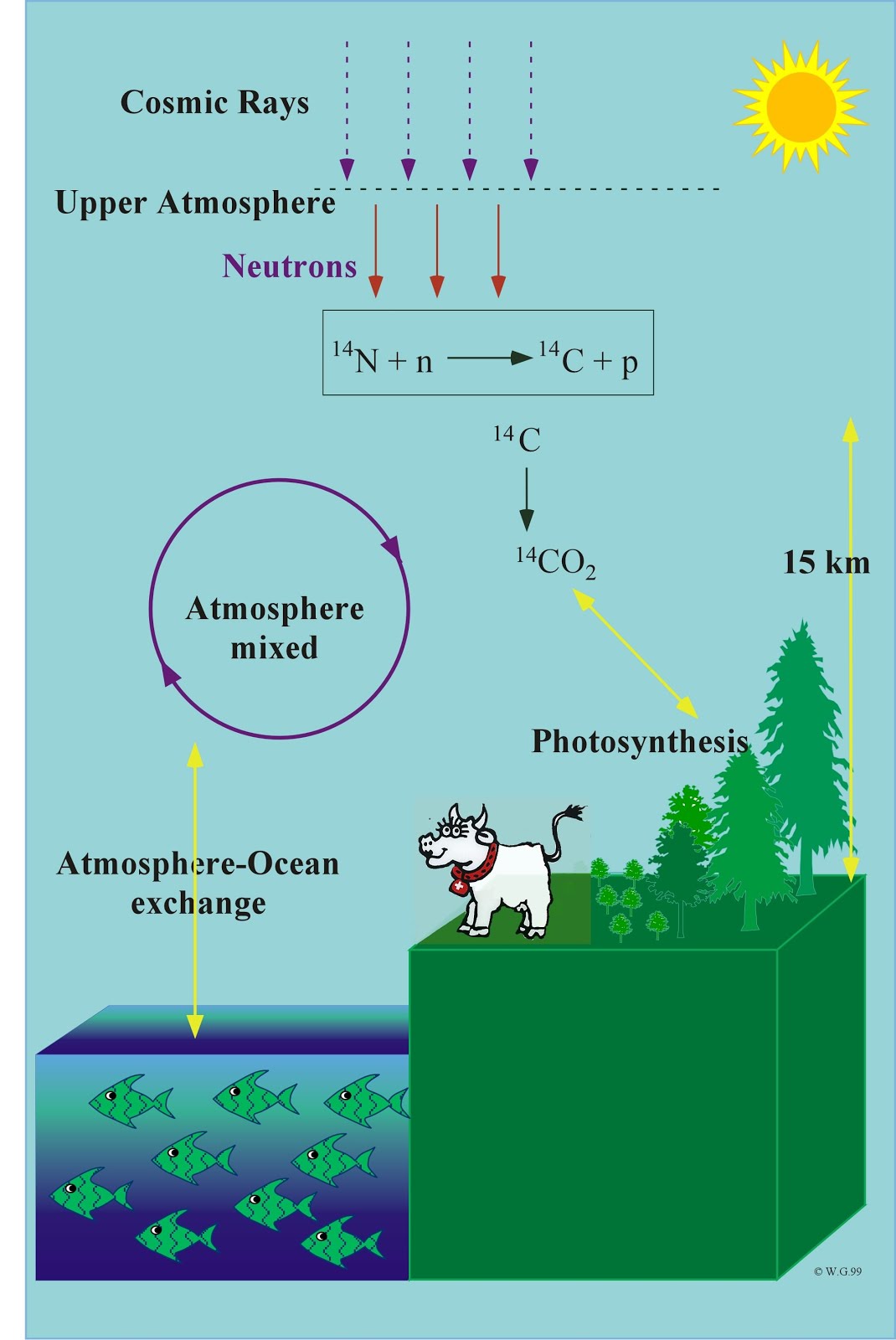
Instead, tissue carbon-14 levels are directly compared to levels in a rapidly changing atmosphere. The forensic use of carbon-14 measurement does not rely on the slow decay process. It has resulted in artificially high levels of carbon-14 in plants and animals living in the past 60 years. The radiocarbon created by the thermonuclear explosions is identical to naturally created radiocarbon, and its route into plant and animal tissues is the same. 
Since then, atmospheric carbon-14 levels have been declining as the radiocarbon is soaked up by the oceans and the biosphere. The Nuclear Test-Ban Treaty of 1963 effectively ended the era of atmospheric testing for most nuclear powers. A large increase in atmospheric carbon-14 occurred when the United States and several other countries tested nuclear weapons aboveground during the 1950s and 1960s (see Figure 1). Natural and anthropogenic fluctuations in environmental radiocarbon levels mean that organisms living in different centuries within the past 500 years can have identical radiocarbon contents.įigure 1: Northern Hemisphere Atmospheric Radiocarbon Levels 1940 to 2003 A.D.įorensic scientists use carbon-14 measurement in a subtly different manner. Objects younger than 500 years old are rarely radiocarbon dated. Radiocarbon measurement can date organic remains up to about 50,000 years old. Knowing the halflife, they back calculate how much time must have passed since the remains had levels identical to living organisms. Archaeologists precisely measure levels of the isotope in organic remains. After an organism dies, its level of carbon-14 gradually declines at a predictable pace, with a half-life of about 5,730 years. It is then incorporated into all living organisms by means of the food chain. The radioactive carbon is taken from the atmosphere and incorporated into plant tissues by plant photosynthesis. Standard carbon-14 testing, as used by archaeologists, is based on the natural process of radioactive carbon formation that results from cosmic ray bombardment of nitrogen in the earth's upper atmosphere. Sidebar to the article Applying Carbon-14 Dating to Recent Human Remains by Philip Bulman with Danielle McLeod-Henning.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
